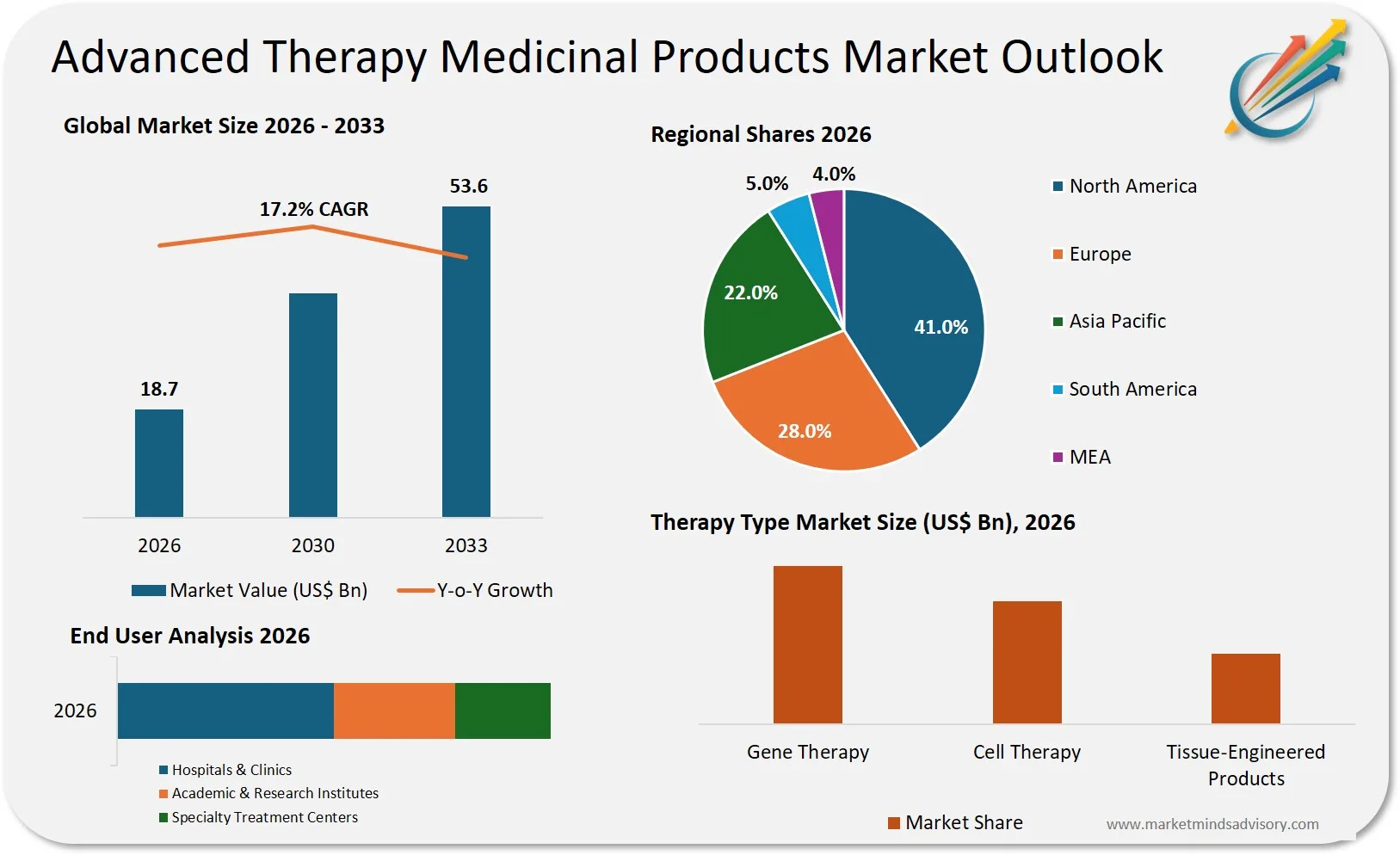
The gap between clinical promise and commercial viability in advanced therapy medicinal products is widening faster than most pharmaceutical executives realize, and it’s creating a bifurcated market where early movers capture disproportionate value while late entrants face structural disadvantages that capital alone cannot overcome.
The advanced therapy medicinal products sector stands at an inflection point where scientific breakthroughs are outpacing business model innovation. While gene therapies, cell therapies, and tissue-engineered products demonstrate unprecedented clinical outcomes, the industry faces a paradox: treatments that can cure diseases in single doses are colliding with reimbursement systems designed for chronic care models. Companies that fail to address this fundamental mismatch are discovering that regulatory approval is merely the beginning of a far more complex commercialization challenge.
Request Report Sample: https://marketmindsadvisory.com/request-sample/?report_id=8107
Why This Market Shift Matters Now
The traditional pharmaceutical playbook is failing in the ATMP space, and the consequences are materializing faster than anticipated. Several approved therapies have been withdrawn not due to safety concerns, but because companies could not solve the economic equation. The cost of goods for autologous cell therapies can exceed $400,000 per patient, while reimbursement negotiations drag on for years. This creates a dangerous gap where approved products cannot reach patients at scale.
What makes this moment particularly critical is the convergence of three forces: a maturing pipeline with over 1,200 cell and gene therapy trials in progress, increasing payer resistance to multi-million dollar single treatments, and manufacturing constraints that prevent economies of scale. Companies that assumed the ATMP market would follow traditional pharmaceutical growth trajectories are now confronting a reality where commercial success requires fundamentally different capabilities in manufacturing, distribution, and value demonstration.
The window for establishing advantaged positions is narrowing. Early entrants are securing exclusive relationships with specialized treatment centers, locking in manufacturing capacity, and shaping reimbursement frameworks that will define the market for the next decade. Organizations still treating ATMPs as an extension of their traditional pharmaceutical business are finding themselves structurally disadvantaged in ways that cannot be quickly remedied.
Structural Shifts Driving the Market
Manufacturing Economics Are Inverting Traditional Pharmaceutical Logic
The shift from centralized to decentralized manufacturing represents more than operational adjustment. It fundamentally changes competitive dynamics. Autologous therapies require patient-specific manufacturing, creating logistical complexity that eliminates traditional scale advantages. Companies investing in point-of-care manufacturing capabilities are discovering they can reduce costs by 40-60% while improving turnaround times from weeks to days. This creates a first-mover advantage in manufacturing infrastructure that becomes increasingly difficult to replicate as capacity constraints tighten.
The emergence of allogeneic approaches offers a different path, enabling off-the-shelf products with traditional manufacturing economics. However, this requires different scientific capabilities and faces distinct regulatory pathways. Organizations must make strategic choices about which manufacturing paradigm to pursue, and these decisions have multi-year implications for capital allocation and competitive positioning.
Reimbursement Models Are Fragmenting Across Geographies
Payer responses to high-cost ATMPs are diverging in ways that create geographic arbitrage opportunities and risks. European markets are experimenting with outcomes-based agreements and installment payments, while U.S. payers remain largely anchored to traditional fee-for-service models with prior authorization barriers. Japan and South Korea are creating fast-track pathways with conditional approvals, accepting higher uncertainty in exchange for patient access.
This fragmentation means companies can no longer pursue uniform global launch strategies. Markets that appear attractive based on epidemiology and GDP may prove commercially unviable due to reimbursement structures, while smaller markets with innovative payment models can generate disproportionate returns. The strategic implication is clear: geographic sequencing and market selection have become as important as clinical development strategy.
The Treatment Delivery Infrastructure Is Becoming a Bottleneck
ATMPs require specialized administration capabilities that exist in only a fraction of healthcare facilities. Cell therapies demand expertise in managing cytokine release syndrome and other acute complications. Gene therapies require pre-treatment protocols and long-term monitoring that most community hospitals cannot provide. This creates a hub-and-spoke model where a limited number of certified centers control patient access.
Companies that secure exclusive or preferred relationships with these centers gain structural advantages in market access. However, this also creates capacity constraints that limit market growth. The number of patients who can receive CAR-T therapy, for example, is constrained not by demand but by the number of beds in qualified treatment centers. Organizations that invest in expanding treatment infrastructure are essentially building moats around their commercial positions.
Where the Real Opportunity Lies
The highest-value opportunities are emerging in therapeutic areas where ATMPs offer not just incremental improvement but fundamental shifts in treatment paradigms. Oncology remains the largest segment, but the real opportunity lies in moving beyond late-line treatments for refractory patients toward earlier intervention where health economic value is clearer. CAR-T therapies approved for second-line diffuse large B-cell lymphoma demonstrate this shift, with reimbursement negotiations proceeding more smoothly when treatments prevent years of costly conventional therapy.
Rare genetic diseases represent a different opportunity profile. Smaller patient populations reduce commercial risk and simplify clinical development, while desperate patients and advocacy groups create pull-through demand that eases reimbursement discussions. However, ultra-rare diseases also mean limited revenue potential, requiring companies to build portfolios rather than betting on single assets.
The emerging frontier lies in chronic diseases where single or infrequent dosing can replace lifetime pharmaceutical regimens. Hemophilia gene therapies that eliminate the need for prophylactic factor replacement demonstrate the economic logic, but success requires convincing payers to make large upfront payments in exchange for long-term savings. Companies that can structure risk-sharing agreements and demonstrate durable efficacy are unlocking markets worth tens of billions in displaced pharmaceutical revenue.
Browse the Complete Report: https://marketmindsadvisory.com/advanced-therapy-medicinal-products-market/
Competitive Dynamics Are Rewarding Integration
The ATMP value chain is consolidating in unexpected ways. Traditional pharmaceutical companies with strong commercial capabilities are acquiring manufacturing and delivery expertise through partnerships and acquisitions. Simultaneously, contract development and manufacturing organizations are moving upstream, offering end-to-end solutions that commoditize aspects of ATMP development.
This creates strategic tension. Vertical integration offers control and margin protection but requires massive capital investment in capabilities outside traditional pharmaceutical core competencies. Outsourcing preserves capital flexibility but risks commoditization and loss of competitive differentiation. Companies in the middle, with partial integration, often face the worst of both worlds: high fixed costs without full control.
The winners are emerging at the extremes. Fully integrated players like Novartis with Kymriah have built end-to-end capabilities from manufacturing through specialized sales forces. At the other end, highly specialized players focusing on narrow therapeutic areas are achieving profitability through focus. The struggling middle consists of companies with broad ATMP pipelines but insufficient scale in any single area to achieve operational efficiency.
The Cost of Delayed Action
Organizations that defer strategic decisions about ATMP capabilities face compounding disadvantages:
- Manufacturing capacity is being locked up through long-term contracts, with lead times for new facilities extending beyond three years. Companies without secured capacity will face production bottlenecks that delay launches and limit commercial potential.
- Treatment center relationships are becoming exclusive, with early movers establishing preferred provider networks that create barriers for later entrants. Breaking into established referral patterns requires significant investment with uncertain returns.
- Reimbursement frameworks are being set based on early products, creating precedents that advantage first movers. Later entrants face price compression and more stringent outcomes requirements as payers gain experience and negotiating leverage.
- Talent with ATMP-specific expertise remains scarce, and competition for experienced professionals is intensifying. Organizations that delay building internal capabilities face escalating talent acquisition costs and longer learning curves.
- Regulatory pathways are evolving based on early submissions, with agencies developing institutional knowledge and expectations. Companies that engage early in regulatory dialogue shape these frameworks, while late entrants must adapt to established paradigms.
What This Means for Decision-Makers
For Pharmaceutical and Biotech Executives
The strategic question is not whether to participate in ATMPs but how to position for sustainable competitive advantage. This requires honest assessment of organizational capabilities and willingness to make bold choices. Half-measures, such as acquiring single assets without building supporting infrastructure, typically destroy value. Success requires either full commitment to building integrated capabilities or strategic focus on specific therapeutic areas where specialized expertise creates defensible positions. The middle ground of opportunistic ATMP investments without strategic coherence is proving to be a value trap.
For Healthcare Systems and Hospital Networks
ATMP administration capabilities are becoming strategic differentiators that influence referral patterns and payer contracts. Health systems that invest in certified treatment centers and specialized care teams are capturing high-value patients and negotiating advantaged reimbursement terms. However, this requires significant upfront investment in infrastructure and training with multi-year payback periods. The decision to build ATMP capabilities must be made in the context of broader strategic positioning and competitive dynamics in local markets.
For Investors and Capital Allocators
Traditional pharmaceutical valuation models break down in the ATMP space. The relationship between clinical success and commercial value is more tenuous, with manufacturing economics, reimbursement strategy, and treatment infrastructure often mattering as much as clinical differentiation. Due diligence must extend beyond pipeline assessment to evaluate operational capabilities and commercial strategy. The highest returns are accruing to investors who can identify companies with not just promising science but also realistic paths to commercial viability at scale.
For Policymakers and Regulators
Current regulatory and reimbursement frameworks were designed for traditional pharmaceuticals and are creating unintended barriers to ATMP adoption. Adaptive licensing pathways, outcomes-based reimbursement models, and infrastructure investment in treatment centers are necessary to realize the public health potential of these therapies. However, policy innovation must balance access with fiscal responsibility and evidence standards. Countries that develop workable frameworks early are attracting investment and clinical development activity, creating competitive advantages in the emerging bioeconomy.
The ATMP market is separating into winners and strugglers faster than most anticipated
The advanced therapy medicinal products sector is not simply another pharmaceutical category. It represents a fundamental shift in how medicines are developed, manufactured, delivered, and paid for. Organizations that recognize this and adapt their strategies accordingly are establishing positions that will generate returns for decades. Those that treat ATMPs as an incremental opportunity within existing business models are discovering that success requires transformation, not adaptation. The decisions made in the next 18 to 24 months will largely determine which companies capture value in this market and which are left struggling to catch up in a race where the leaders are pulling away.
About Company
At Market Minds, we’re more than just consultants—we’re partners in your journey to growth and success. We combine deep industry expertise with cutting-edge research to uncover insights that truly matter, helping you navigate challenges and seize opportunities with confidence. Whether it’s adapting to market shifts, exploring new revenue streams, or staying ahead of emerging trends, our focus is always on delivering tailored solutions that drive real results. With us, you’re not just getting advice—you’re gaining a trusted team dedicated to your success, every step of the way.
Contact Us
Market Minds Advisory
86 Great Portland Street, Mayfair,
London, W1W7FG,
England, United Kingdom
Phone: +44 020 3807 7725
Email: [email protected]
Website: https://marketmindsadvisory.com/