Navigating a cancer diagnosis often involves deciphering a complex web of medical terminology, treatment options, and regulatory landscapes. Patients frequently encounter terms like "integrative" and "alternative" used interchangeably, yet these approaches represent fundamentally different paths in oncology. Understanding this distinction is a must for making right safety decisions, especially when exploring advanced immunotherapies abroad.
This article clarifies these definitions and examines the regulatory divide, using the comparison of NK Cell Therapy in Mexico vs United States to illustrate how location dictates access to emerging treatments.
Experimental Immunotherapy Access - Defining the Core Approaches
To understand the treatment landscape, one must first distinguish between the methodologies. Integrative cancer treatment combines conventional medicine like chemotherapy, radiation, and surgery with evidence-based complementary therapies. The goal is to support the patient’s physiology, reduce side effects, and improve quality of life without rejecting standard-of-care protocols. It is a collaborative model where oncologists and holistic practitioners work in tandem.
In contrast, alternative cancer treatment typically refers to therapies used instead of conventional medicine. These are often unproven as standalone cures in clinical trials and are not part of the standard oncology toolkit in the United States.
However, the line between "experimental" and "alternative" often shifts depending on the country of treatment. This is nowhere more apparent than in the field of immunotherapy, specifically when comparing NK Cell Therapy in Mexico vs the United States.
The Regulatory Divide: FDA vs. COFEPRIS
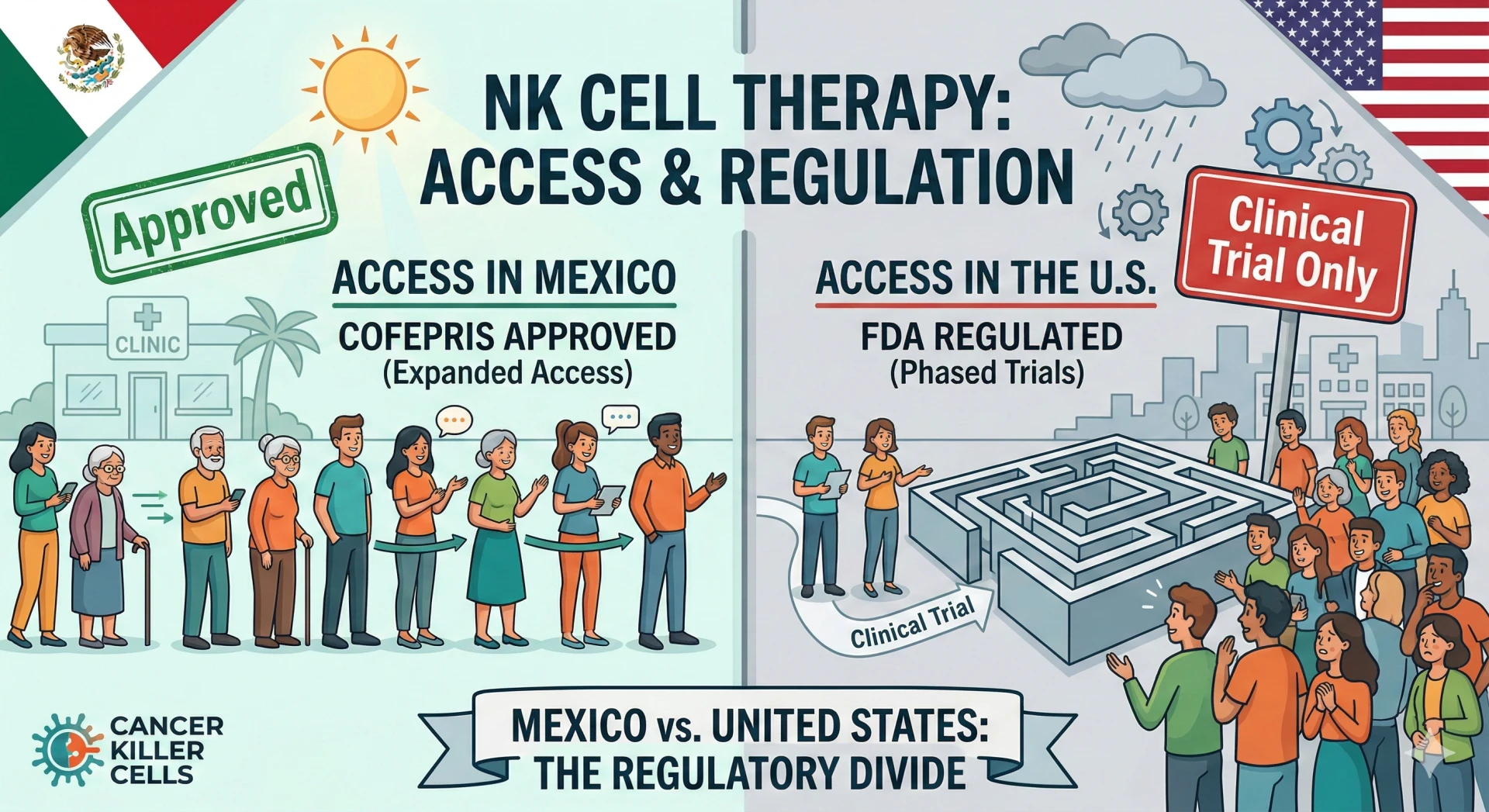
The United States and Mexico operate under vastly different regulatory frameworks, which directly influences which treatments are considered "integrative" versus "alternative." In the U.S., the Food and Drug Administration (FDA) maintains a rigorous, multi-phase approval process. As of early 2026, the NK cell therapy FDA approval status remains restrictive; while numerous Investigational New Drug (IND) applications are active, full approval for commercial marketing of NK cell therapies is limited to specific, narrow indications or strictly confined to clinical trials.
Mexico, conversely, regulates its medical sector through COFEPRIS (Federal Committee for Protection from Sanitary Risks). NK cell therapy regulations Mexico are often described as more flexible regarding the application of regenerative medicine. While Mexican law still requires safety and efficacy standards, the pathway for clinics to offer cell-based therapies is distinct.
NK Therapy legal status Mexico allows licensed facilities to administer treatments that are still in Phase I or II trials north of the border. This regulatory divergence means a therapy considered "experimental" in the U.S. might be legally available as a therapeutic option in Mexico.
Immunotherapy Approval Process: A Case Study in Access
Natural Killer cells are the immune system’s first line of defense, capable of identifying and eliminating cancer cells without prior sensitization. The primary difference in NK Cell Therapy in Mexico vs United States lies in the types of cells available and the methods of administration.
Clinical Trial Requirements and "Minimal Manipulation"
In the United States, clinical trial requirements United States are stringent. The FDA generally enforces a "minimal manipulation" rule for human tissue. This means that if cells are significantly processed or expanded in a lab to increase their numbers, they are classified as drugs. This classification triggers a requirement for years of pre-market testing. Consequently, most U.S. patients can only access high-dose, expanded NK cells if they qualify for a specific clinical trial.
Expanded Access in Mexico.
Mexican clinics, operating under regenerative medicine regulations, frequently offer "expanded" allogeneic NK cell therapies. These protocols involve culturing cells in a laboratory to reach counts in the billions—numbers difficult to achieve under the strict "minimal manipulation" constraints faced by U.S. clinics outside of trials.
For a patient seeking NK cell clinic Mexico, the choice often comes down to access: the ability to receive a high-dose immunotherapy immediately versus waiting for trial enrollment eligibility.
Immunotherapy Regulations USA and Patient Safety
The distinction between integrative and alternative care becomes crucial when leaving the U.S. healthcare system. The immunotherapy regulations in the USA are designed to maximize safety through slow, deliberate data collection. The trade-off is time. Patients with aggressive malignancies often feel they cannot afford the years it takes for FDA approval NK cell therapy to materialize for their specific condition.
However, the "alternative" label in Mexico does not necessarily imply a lack of science. Many Mexican clinics utilize protocols similar to those in U.S. trials but offer them commercially. The risk lies in vetting. Experimental cancer treatment laws in Mexico are robust on paper, but enforcement can vary.
Patients must rigorously verify that a facility holds valid COFEPRIS licenses for cell banking and administration. In this context, integrative care involves retaining a U.S. oncologist to monitor progress while accessing these external therapies, rather than abandoning conventional care entirely.
Evaluating the Risks and Benefits
When considering cancer immunotherapy in Mexico, patients must weigh the potential for advanced access against the protection of FDA oversight.
* United States: High safety assurance, financial coverage (if in a trial), but strictly limited availability and lower cell counts for non-trial patients.
* Mexico: Immediate access to expanded cell counts and broader inclusion criteria, but requires out-of-pocket payment and diligent self-verification of clinic credentials.
Ultimately, the decision rests on the patient's specific medical needs and risk tolerance. Integrative medicine encourages using all available tools. For some, this means combining U.S. standard care with lawful, advanced immunotherapies available internationally.
Summing Up:
The difference between integrative and alternative cancer treatment is not just about the therapy itself, but where and how it is administered. While the U.S. categorizes many cell therapies as experimental due to strict clinical trial requirements United States, other jurisdictions view them as viable medical treatments. By understanding the regulatory gap defining the true purpose of medical travel for cancer treatment, patients can move beyond confusing terminology.
The most effective approach often lies in a truly integrative strategy: leveraging the safety of conventional diagnostics while thoughtfully exploring advanced immunotherapies that regulations may not yet permit at home.
Contact Cancer Killer Cells for a prompt and effective approval process.