In today’s competitive and highly regulated industries, maintaining quality standards is more than a compliance requirement—it’s a critical factor for business success. Organizations across pharmaceuticals, life sciences, manufacturing, and healthcare increasingly rely on Corrective and Preventive Action (CAPA) software to manage quality issues efficiently.
Qualityze Corrective and Preventive Action Software is a cloud-based, enterprise-grade solution designed to automate, streamline, and track CAPA processes, helping businesses reduce risks, ensure compliance, and improve operational efficiency.
What Is Corrective and Preventive Action (CAPA) Software?
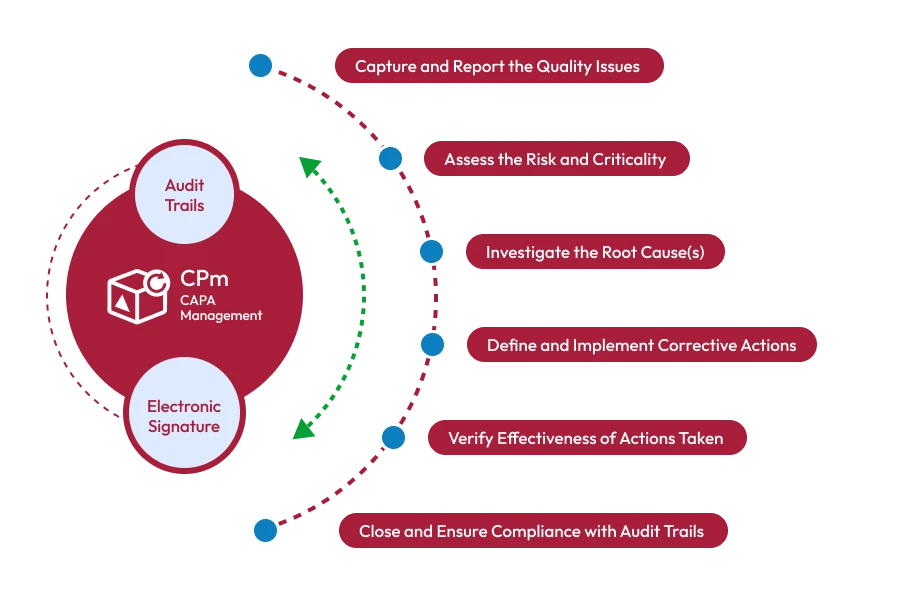
CAPA software is a quality management solution that automates the identification, investigation, and resolution of quality issues. It ensures that organizations can:
- Correct existing problems through corrective actions.
- Prevent future issues through preventive actions.
- Maintain complete traceability of all actions and approvals.
Qualityze CAPA Software integrates seamlessly with other Quality Management System (QMS) modules such as:
- Nonconformance Management
- Audit Management
- Document Management
- Change Management
This ensures that all quality events are managed in a centralized, compliant system.
Key Features of Qualityze CAPA Software
1. Automated CAPA Workflows
- Initiate CAPA from nonconformance reports, audits, or customer complaints.
- Assign tasks to responsible teams with deadlines and priority levels.
- Automate reminders and escalation to ensure timely resolution.
2. Root Cause Analysis (RCA)
- Conduct structured investigations using 5-Why Analysis and Fishbone Diagrams.
- Document findings to prevent recurrence and maintain audit-ready records.
3. Regulatory Compliance
- Compliant with FDA 21 CFR Part 11, ISO 9001, ICH Q10, and GMP standards.
- Electronic signatures and audit trails ensure data integrity and accountability.
4. Integrated Dashboard
- Real-time visibility of CAPA status, overdue tasks, and effectiveness checks.
- Customizable dashboards and reports for audits and management review.
5. Corrective and Preventive Action Tracking
- Track the CAPA lifecycle from initiation to closure.
- Evaluate preventive measures to ensure recurring issues are avoided.
- Maintain complete traceability of all actions and approvals.
6. Configurable Templates and Forms
- Customize CAPA workflows, forms, notifications, and reports.
- Adapt the software to unique organizational processes without coding.
7. Cloud-Based and Secure
- Hosted on the Salesforce platform, offering enterprise-grade security.
- Accessible anytime, anywhere, ideal for multi-site organizations.
Benefits of Implementing Qualityze CAPA Software
- Improved Compliance: Adheres to global regulatory standards, reducing audit risks.
- Faster Issue Resolution: Automates CAPA workflows and root cause analysis for timely corrective action.
- Operational Efficiency: Reduces manual errors and improves productivity.
- Data-Driven Insights: Provides real-time dashboards for better decision-making.
- Continuous Improvement: Identifies recurring trends and drives process enhancements.
- Centralized Quality Operations: All CAPA-related data is stored in a single, secure system.
Industries That Benefit from Qualityze CAPA Software
IndustryUse CasePharmaceuticalsMaintain GxP compliance, track deviations, and prevent recalls.Medical DevicesManage CAPAs to meet FDA and ISO regulations.Life SciencesEnsure quality in laboratory and clinical operations.ManufacturingReduce operational defects and improve process quality.Food & BeverageTrack quality issues, prevent recurrence, and ensure food safety.
Why Qualityze Is a Leading CAPA Software Solution
- Cloud-Based & Scalable: Accessible globally, supporting multi-site operations.
- Integrated QMS: Works with nonconformance, audits, document control, and training modules.
- Customizable Workflows: Configurable forms and processes to match organizational needs.
- Data-Driven Decision Making: Advanced analytics help prevent recurring issues and optimize quality processes.
- Regulatory Compliance Ready: Built to meet international standards for audit-ready quality management.
Conclusion
Qualityze Corrective and Preventive Action Software is the ultimate solution for organizations seeking a comprehensive, cloud-based CAPA system. By automating workflows, maintaining compliance, and providing actionable insights, Qualityze empowers businesses to resolve quality issues efficiently, prevent future problems, and foster a culture of continuous improvement.
For enterprises aiming to enhance product quality, reduce compliance risks, and improve operational efficiency, Qualityze CAPA Software is a reliable, scalable, and secure choice.