The Role of the Ames Test in Predicting the Genotoxicity of Impurity Compounds in Drugs
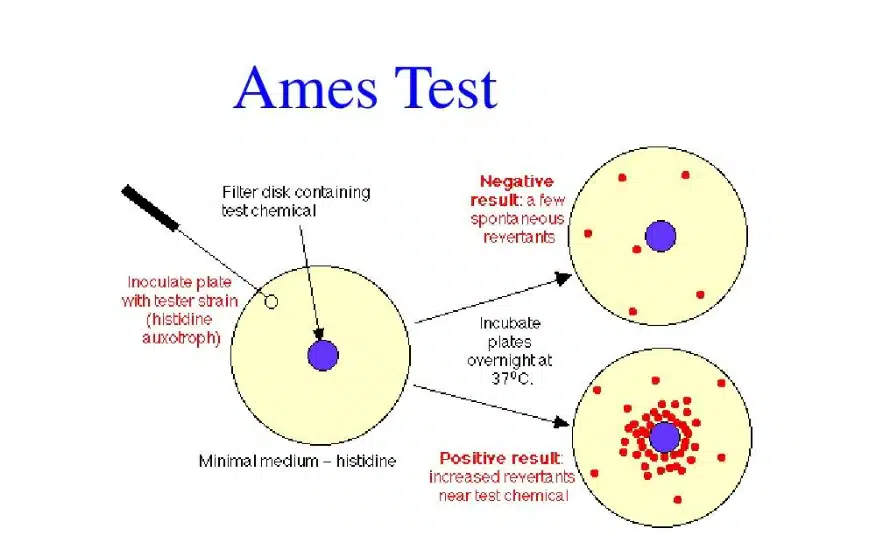
Many impurities in drugs can produce toxic and side effects, which is a crucial factor affecting the safety and effectiveness of drugs, especially genotoxic impurities that can cause serious harm to human health with minimal exposure. Therefore, to ensure API quality and preparations, Genotoxic impurities are a class of impurities that must be paid attention to in the drug synthesis process. Genotoxic impurities from chemical reagents, chemical synthesis, and reaction are involved in all aspects of the drug synthesis process and the subsequent stability and possible degradation of the drug, which is a complex process. For potential carcinogenic or mutagenic substances and impurities with warning structures, the Ames test can generally be used to test the mutagenic ability of impurities.
The Ames test is short-term in vitro test method for preliminary screening of the mutagenicity of chemical substances by using the properties of the reverse mutation of the histidine auxotrophic mutant strain of Salmonella typhimurium. Through the Ames test, the genotoxicity of impurities can be defined, the genotoxicity hazard identification can be carried out, and whether the target compound can cause damage to the genetic material can be identified. The exposure can be further controlled to reduce the risk.
1. Classification of genotoxic impurities
Toxicological concern threshold TTC limits are used as acceptable limits for genotoxic impurities. According to the PhRMA model, genotoxic impurities from chemical reactions are divided into five categories.
(1) Impurities known to be genotoxic and carcinogenic, such as aflatoxins, N. Compounds such as nitroso species face a very high risk of genotoxicity even when ingested in amounts below the TTC value. To avoid such impurities, even the drug synthesis process must be changed.
We boast professional teams and practical experience in drug safety evaluation and can promise high-quality data and fast turnaround time to support various drug safety evaluation. Our toxicology research can be carried out according to non-GLP or GLP standards. Our research platform has been rated as Shanghai R&D Public Service Platform.
(2) Impurities known to have genotoxicity but unknown carcinogenicity. The bacterial mutation test was positive, but no rodent carcinogenicity data.
(3) Impurities with a warning structure but not related to the final API structure. For impurities with alert structures), adequate controls can be applied, or an Ames test can be performed on the impurity alone. If the Ames test result is negative, the impurity is considered non-genotoxic, and no further testing is required. If the Ames test result is positive, further hazard analysis or control measures should be carried out.
For Type 2 and Type 3 impurities, risk assessment and necessary doping (marking) and cleaning operations should be carried out, and quality control should be performed by tracking, inferring, and using the TTC threshold adjustment mechanism.
(4) Impurities with warning structures related to API. Suppose the impurity has a warning structure similar to the API, and the Ames test result of the API is negative or previous studies have confirmed no genotoxicity. In that case, the impurity can be considered non-genotoxic.
(5) Impurities with no warning structure or sufficient evidence to prove no genotoxicity. Type 4 and Type 5 impurities can be treated as general; the content should be less than 0.1%. Solving the impurity problem, making impurities no longer complicated and troublesome, will help the success of drug synthesis process research. Medicilon’s process chemistry team has rich scientific management experience and a rigorous scientific exploration spirit. It can provide customers with mature one-stop services from drug discovery and synthesis to process research.
For example, some researchers use the Ames test to investigate whether dapagliflozin is mutagenic. The method uses the standard plate incorporation method and applies histidine auxotrophic Salmonella typhimurium TA97, TA98, TA100, TA102, and TA1535 to carry out a reverse mutation test on canagliflozin. Results In the dose range of 31.25-2 000 μg/dish, the dapagliflozin Ames reverse mutation test results were negative under similar conditions with and without rat liver microsomal enzyme S9. The study found that under the test conditions, dapagliflozin had no mutagenicity.
In most cases, methods limited to in vitro studies of impurities, such as the Ames test, chromosomal aberration tests, and toxicological evaluation of genotoxic impurities, are not suitable for determining acceptable intake levels of impurities. That is to say, it is inappropriate to calculate the “safety factor” of impurities based on in vitro data such as the Ames test and then determine the acceptable limit. In addition, even if a negative result is obtained for carcinogenicity and genotoxicity studies with APIs containing lower (ppm level) impurity levels, it is not enough to ensure the rationality of the impurity limit because this test method lacks the necessary sensitivity. Some highly mutagenic and carcinogenic substances are tested together with the drug substance because, at deficient exposure levels, there is a high probability that they will not be detected because they are below the detection limit. So, if it is recognized that there is no “unacceptable risk” of a genotoxic impurity at very low levels, then a practical approach can be taken to control this impurity.
