Regulatory Services For biological Product in India
India has rapidly emerged as a significant player in the global pharmaceutical landscape, with a particular emphasis on biological products. The country's robust scientific talent, cost-effective manufacturing capabilities, and a large patient pool make it an attractive hub for the development and production of biologics, including vaccines, biosimilars, monoclonal antibodies, and advanced therapy medicinal products. However, successfully navigating this dynamic environment requires a deep understanding of the sophisticated regulatory framework governing Regulatory Services for Biological Products in India.
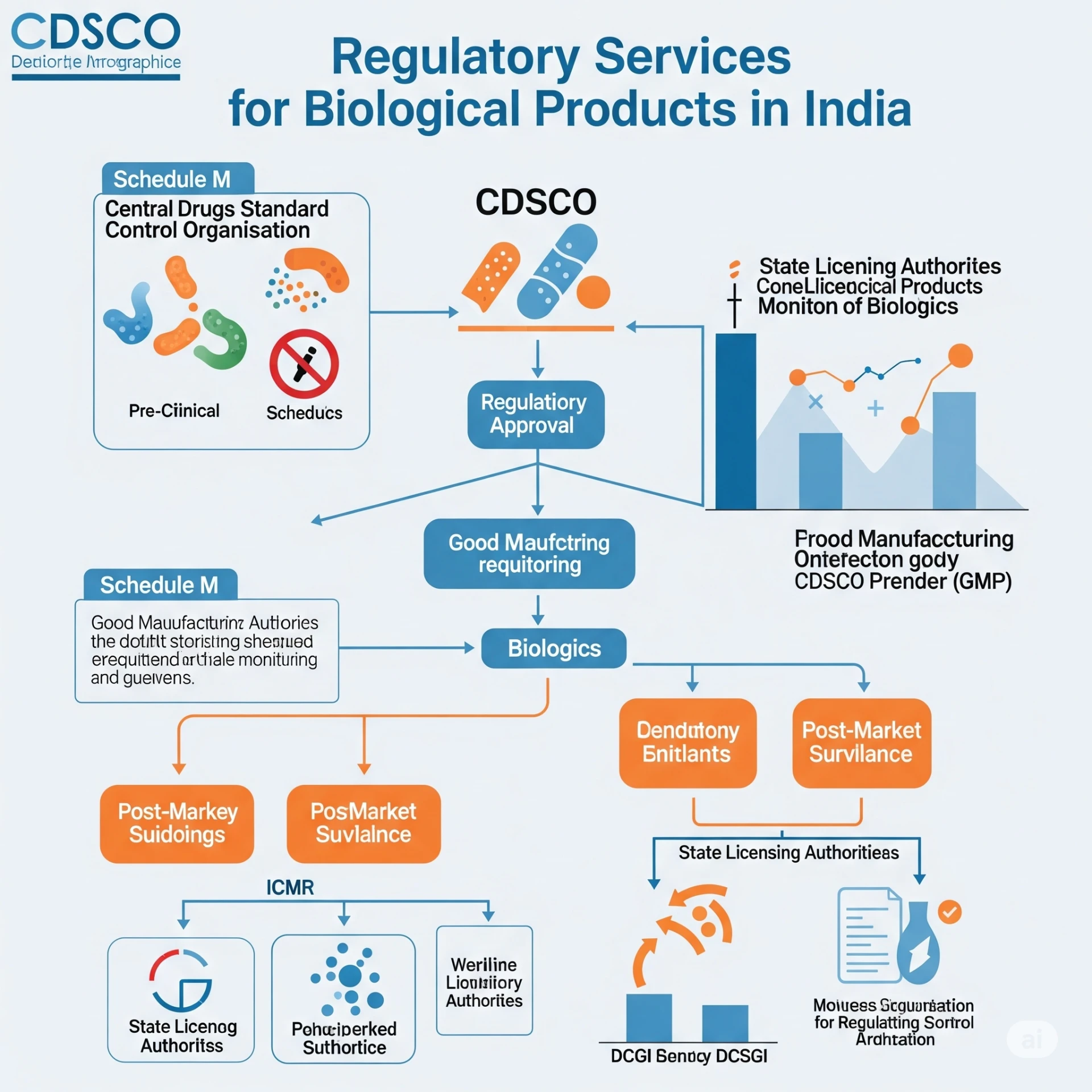
The Apex Regulatory Authority: CDSCO
At the helm of drug regulation in India is the Central Drugs Standard Control Organization (CDSCO), under the Ministry of Health & Family Welfare. Headed by the Drug Controller General of India (DCGI), CDSCO is the primary authority responsible for the approval of new drugs, including all biological products, and regulating clinical trials. Its mandate extends to ensuring the safety, efficacy, and quality of these complex therapies across their lifecycle, from development to post-market surveillance.
Key Regulations and Guidelines
The overarching legal framework for drugs in India is the Drugs and Cosmetics Act, 1940, and Rules, 1945. However, given the unique nature of biological products, specific guidelines and rules have been formulated to address their complexity and ensure their safe introduction into the market.
A pivotal piece of legislation is the New Drugs and Clinical Trials Rules, 2019. These rules provide a comprehensive regulatory framework for new drugs, investigational new drugs, and specifically address various categories of biological products. They aim to streamline the approval process, enhance transparency, and align Indian regulations with international best practices (like ICH guidelines). For biological products, these rules delineate requirements for pre-clinical studies, clinical trials (Phases I, II, III), and marketing authorization.
Beyond the 2019 Rules, the "Guidelines on Similar Biologics", jointly issued by CDSCO and the Department of Biotechnology (DBT), are crucial. These guidelines specifically lay down the regulatory pathway for biosimilars, emphasizing the need for extensive comparability exercises to demonstrate similarity in terms of quality, safety, and efficacy to an approved reference biologic.
Furthermore, other specific guidelines are applicable:
- Recombinant DNA Safety Guidelines, 1990: Addresses genetically engineered organisms.
- Guidelines for generating preclinical and clinical data for rDNA vaccines, diagnostics, and other Biologicals, 1999: Provides specific data requirements.
- Rules for the manufacture, use, import, export and storage of hazardous microorganisms/genetically engineered organisms or cells, 1989 (Rules, 1989): Notified under the Environment (Protection) Act, 1986, these rules involve inter-ministerial committees.
Multi-Agency Oversight for Biologics
The regulation of biological products in India involves a multi-agency approach, reflecting their complex nature and potential environmental implications. Beyond CDSCO, key bodies include:
- Review Committee on Genetic Manipulation (RCGM): Functioning under the Department of Biotechnology (DBT), RCGM is responsible for authorizing R&D activities, exchange of genetically engineered cell banks, and reviewing data up to preclinical evaluation, particularly for recombinant DNA (rDNA) derived products.
- Genetic Engineering Appraisal Committee (GEAC): Under the Ministry of Environment, Forest, and Climate Change (MoEF&CC), GEAC is a statutory body that reviews applications and grants approvals for activities where the final drug product contains genetically modified organisms (GMOs) or living modified organisms (LMOs).
- Institutional Biosafety Committees (IBSCs): Required at the institutional level for anyone handling hazardous microorganisms or genetically engineered organisms, IBSCs ensure on-site biosafety and provide initial review for applications to RCGM.
The Approval Process for Biological Products
The approval process for biological products in India is comprehensive and multi-staged:
- Pre-clinical Development and Comparability Studies: For new biologics, extensive pre-clinical studies (in vitro and in vivo) are required. For biosimilars, rigorous analytical and functional characterization to demonstrate "similarity" to the reference product is paramount.
- Regulatory Review by RCGM/GEAC (if applicable): For products involving genetic engineering, initial clearances from RCGM and potentially GEAC are required before clinical trials.
- Clinical Trial Approval: An Investigational New Drug (IND) application is submitted to CDSCO for approval to conduct human clinical trials. The New Drugs and Clinical Trials Rules, 2019, aim for faster approvals (e.g., 30 days for domestic trials, 90 days for global trials) and include provisions for waiver of local clinical trials if the drug is approved and marketed in certain recognized countries (e.g., US, EU, UK, Canada, Australia, Japan) without significant safety concerns or metabolic differences in the Indian population.
- Clinical Trials (Phase I, II, III): Trials are conducted as per Good Clinical Practice (GCP) guidelines, with oversight from Ethics Committees registered with the Department of Health Research (DHR).
- Marketing Authorization Application (MAA): Upon successful completion of clinical trials, a comprehensive dossier (preferably in CTD format) is submitted to CDSCO. This dossier includes detailed information on quality (CMC), non-clinical and clinical data, and pharmacovigilance plans.
- Post-Marketing Surveillance: Upon approval, companies must establish a robust pharmacovigilance system, including a Qualified Person for Pharmacovigilance (QPPV), maintaining safety databases, and submitting Periodic Safety Update Reports (PSURs) to CDSCO.
Challenges and Future Outlook
While India's regulatory framework for biological products has matured significantly, challenges persist. These include the need for continuous harmonization with evolving global standards, managing the complexity of advanced therapies like gene and cell therapies, and ensuring consistent interpretation and application of guidelines across all stakeholders.
However, the future for Regulatory Services for Biological Products in India remains promising. The government's push for "Make in India" initiatives, increasing investment in biopharmaceutical R&D, and the continuous refinement of regulatory pathways are positioning India as a global leader in accessible and high-quality biological medicines. Strategic regulatory services, offering expertise in dossier preparation, clinical trial management, and post-market compliance, are critical for pharmaceutical companies looking to successfully introduce biological products into this rapidly expanding market.